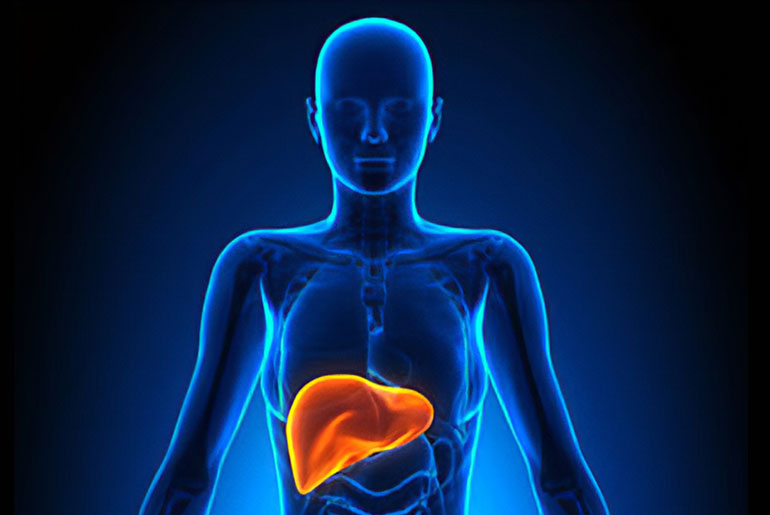
Liver fibrosis is a condition where damaged liver cells are replaced by scar tissue, potentially leading to cirrhosis, liver failure, and even liver cancer. It results from cumulative scarring that restricts blood flow to the liver over time. A group of researchers, led by David A. Brenner, M.D., and Tatiana Kisseleva, M.D., Ph.D., has explored the origin and fate of liver myofibroblasts, the cells responsible for forming liver scar tissue. Their research suggests that liver fibrosis can be reversed under the right conditions.
In normal, healthy livers, myofibroblasts are not present. They become activated in response to chronic liver injuries, such as excessive alcohol consumption. An inflammatory response triggers the activation of hepatic stellate cells (aHSC), which transform from relatively scarce, quiescent cells storing vitamin A into proliferative myofibroblasts that produce scar-forming proteins.
“Liver fibrosis is quite common, in part because it can be caused by a number of things that damage the organ but result in scarring rather than healthy repair,” stated Brenner, a gastroenterologist and noted liver disease researcher. “Everything from viral hepatitis to excessive alcohol consumption to non-alcoholic fatty liver disease (NAFLD), which is extremely common in the US.”
These myofibroblasts create non-functional scar tissue, gradually thickening and coalescing, which can obstruct and damage the liver, leading to life-threatening conditions. Researchers are exploring a therapeutic approach focused on disrupting the formation of hepatic myofibroblasts while addressing the underlying cause of liver damage, such as viral infections or excessive drinking. The idea is that by doing so, aHSCs will revert to their dormant state, and liver fibrosis may regress.
“There are many causes of liver fibrosis and thus, there are no effective therapies currently because most efforts have focused on single targets when the nature of chronic liver injury is incredibly complex,” Brenner stated.
“That means zeroing in on the factors that activate hepatic stellate cells,” stated Brenner, signaling pathways that turn them into scar tissue factories.
Recent studies in mouse models and humans have begun to shed light on the cellular and molecular mechanisms behind this process. Advanced tools like spatial transcriptomics, which can provide comprehensive insights into tissue organization and architecture at the single-cell or subcellular level, are expected to further advance our understanding of liver fibrosis and its potential treatments. This research is particularly relevant as non-alcoholic fatty liver disease (NAFLD), which can progress to more severe forms like nonalcoholic steatohepatitis (NASH) with fibrosis, affects a substantial portion of the global population.
Disclaimer:
The information contained in this article is for educational and informational purposes only and is not intended as a health advice. We would ask you to consult a qualified professional or medical expert to gain additional knowledge before you choose to consume any product or perform any exercise.